 |
Aromatase inhibitors and Adjuvant Treatment of Breast Cancer |
Aromatase inhibitors and Adjuvant Treatment of Breast Cancer. First study to give benefit and safety data for 5 years of treatment.
The 27th Annual San Antonio Breast Cancer Symposium, December 8 - 11, 2004 emphasized a broad spectrum of aspects of clinical trials, including perhaps more than ever before not only treatment effectiveness, but also treatment safety and Quality of Life (QOL). One of the special aspects of aromatase inhibitors, a new class of treatment options for the adjuvant therapy of breast cancer, is that they have different safety issues than tamoxifen. Of the questions to be answered about aromatase inhibitors is not only questions about their effectiveness but also their safety and QOL advantages or disadvantages compared to tamoxifen.
The interest and importance of aromatase inhibitors was reflecting by the fact that the first lecture at the Symposium was a review of aromatase inhibitors given by Dr. Paul Goss. Reflecting on how initial work with aromatase inhibitors in the treatment of metastatic disease has led to advances in adjuvant therapy, he said "A clue to the fact that these inhibitors were going to turn out to be dynamite, in the adjuvant setting, was the extraordinary increase in time-to-progression. When compared to Tamoxifen, a more durable remission occurred."
Aromatase inhibitors work by stopping the enzyme aromatase from converting androgens (male hormones secreted in the adrenal glands and other body tissues) into estrogen. In postmenopausal women, this is thought to be a primary source of estrogen. Aromatase is made in the some breast cancers, which might allow them to supply themselves with estrogen. Aromatase inhibitors block aromatase from producing estrogen in the adrenal glands and other places in postmenopausal women. These may keep estrogen from feeding tumors. The three aromatase inhibitors currently in use are: Arimidex (anastrozole) and Femara (letrozole) which are non-steroidal aromatase inhibitor, and Aromasin (exemestane) is which is a steroidal aromatase inhibitor.
Dr. Goss reviewed the progress that has been made in understanding how aromatase inhibitors might be used in the adjuvant therapy of breast cancer. Studies have shown that tamoxifen that using aromatase inhibitors instead of, or after 2 or 5 years of tamoxifen, are superior to tamoxifen alone, but which aromatase strategy is best is still uncertain. Interestingly using tamoxifen together with the aromatase inhibitor, anastrozole, at the same time was less effective than using an aromatase inhibitor, anastrozole, alone.
The ending quote from Dr. Goss' talk was, "Fully exploiting aromatase inhibition will likely be a cornerstone of reducing breast cancer morbidity and mortality in the future."
General Session 1 The ATAC (Arimidex, Tamoxifen, Alone or in Combination) completed treatment analysis: Anastrozole demonstrates superior efficacy and tolerability compared with tamoxifen
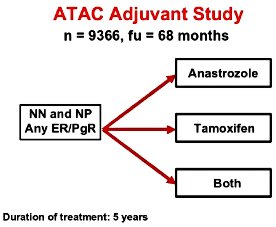 The first talk reporting on clinical research was given by Dr. Anthony Howell, of the University of Manchester, Manchester, United Kingdom. He reported on a large international clinical study evaluating the adjuvant use of anastrozole. What was particularly important about this presentation was that ATAC is the first study to report on fairly long term, greater than 5 years, of adjuvant treatment with aromatase inhibitors. The ATAC Adjuvant Study diagram shows the treatment plan in which 9,366 women were randomly assigned to 5 years of one of three treatment strategies. The treatment strategies were 5 years of tamoxifen, 5 years of the aromatase inhibitor of anastrozole, or both together. An earlier analysis of the ATAC study after about 3 years showed that the combination of both together (anastrozole and tamoxifen) was more less safe and less effective than anastrozole alone, so that this combined therapy was stopped and thus this presentation in 2004 concentrated on just the women that got either tamoxifen or who got anastrozole.
The first talk reporting on clinical research was given by Dr. Anthony Howell, of the University of Manchester, Manchester, United Kingdom. He reported on a large international clinical study evaluating the adjuvant use of anastrozole. What was particularly important about this presentation was that ATAC is the first study to report on fairly long term, greater than 5 years, of adjuvant treatment with aromatase inhibitors. The ATAC Adjuvant Study diagram shows the treatment plan in which 9,366 women were randomly assigned to 5 years of one of three treatment strategies. The treatment strategies were 5 years of tamoxifen, 5 years of the aromatase inhibitor of anastrozole, or both together. An earlier analysis of the ATAC study after about 3 years showed that the combination of both together (anastrozole and tamoxifen) was more less safe and less effective than anastrozole alone, so that this combined therapy was stopped and thus this presentation in 2004 concentrated on just the women that got either tamoxifen or who got anastrozole.
The following table shows reduction of the risk for women who got anastrozole compared to a tamoxifen. It is important to know that these reductions in the risk of relapse as percentages of the risk for a woman who would have gotten tamoxifen. Thus if a woman that a risk of relapse of 20% if she got tamoxifen alone, then if she got anastrozole she would have a 17% reduction in this risk (17% of 20%) or 3.4% reduction in the risk of relapse. It is important to note that although there appeared to be a reduction in the risk of dying of breast cancer this result was not yet statistically certain. An interesting aspect of the additional benefit for anastrozole was that it seemed biggest for patients who were estrogen receptor positive and progesterone receptor negative and less so for patients who were positive for both the estrogen and progesterone receptors.
| Reduction in Risk | |
| Risk of relapsing | 17% |
| Risk of developing metastatic breast cancer | 16% |
| Risk of dying of breast cancer | 13% |
An analysis of side effects of anastrozole as compared to tamoxifen showed that patients who got tamoxifen had a higher risk of blood clots and endometrial cancer, while patients who go anastrozole had a higher risk of bone fractures. Clearly if women are to get anastrozole they will need to have bone loss (osteoporosis) monitored and treated. Additional research about the protection of bone was presented at the symposium.
At the Satellite Symposium on Saturday, December 11, 2004
Breaking the 5-year SERM Barrier: Perspectives on Clinical Application of New Data & Strategies in the Adjuvant Setting. The speakers said results with Aromatase Inhibitors were an important advance, which we hope further results will show to be capable of saving lives and causing a drop in the death rate from Breast Cancer.
| Respectfully submitted by: | Alexandra Andrews CancerLynx.com |
Claudia Kruggel Breast Cancer Today |
| Medical input by: | Peter Ravdin, MD, PhD Associate Professor, Department of Medicine Division of Medical Oncology of Texas Health Science Center San Antonio, Texas |
|
Acknowledgement:
We wish to thank the San Antonio Breast Cancer Symposium Advocate Program for their support of this article
|
|
|